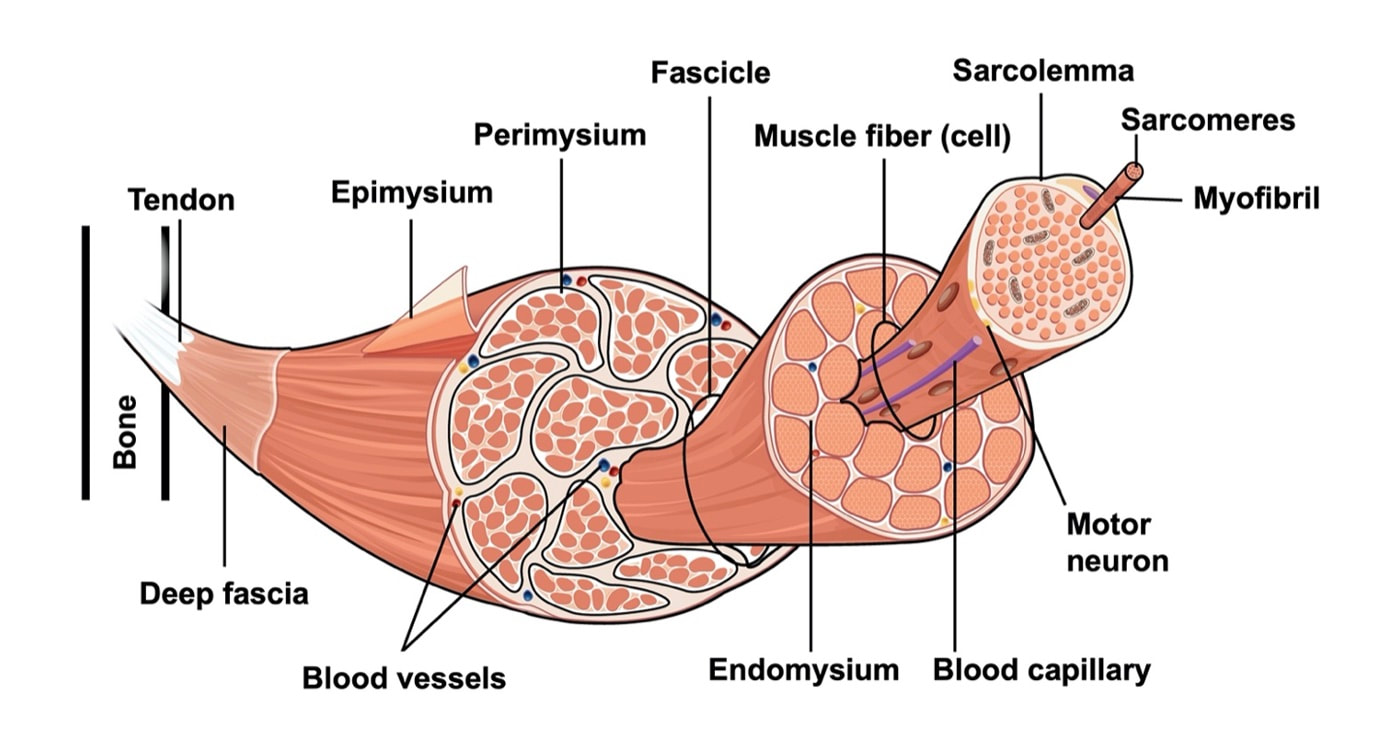
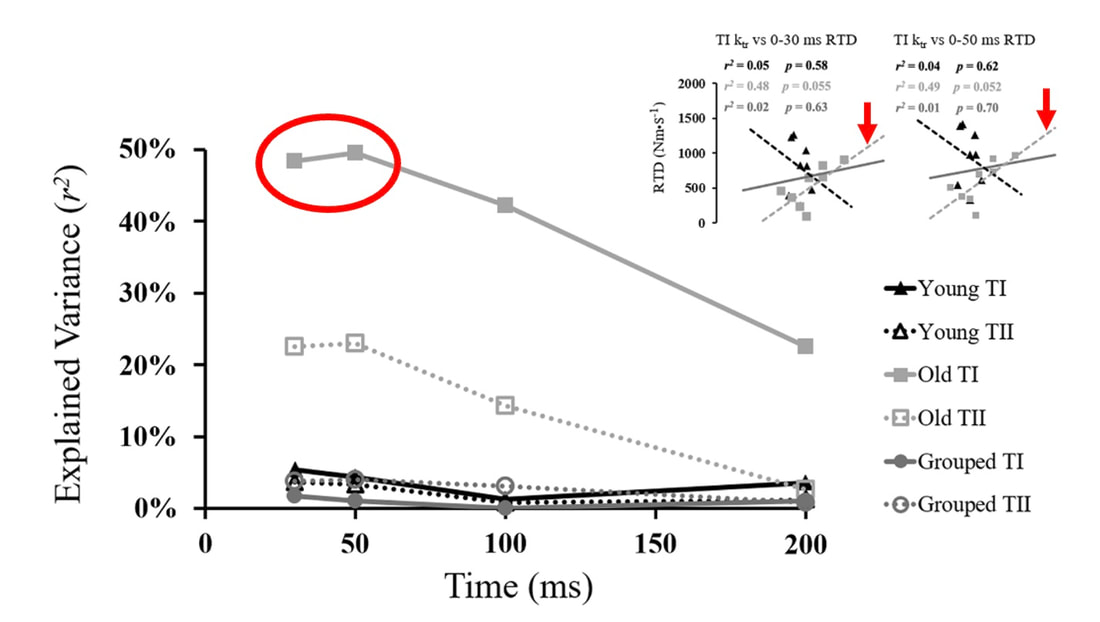
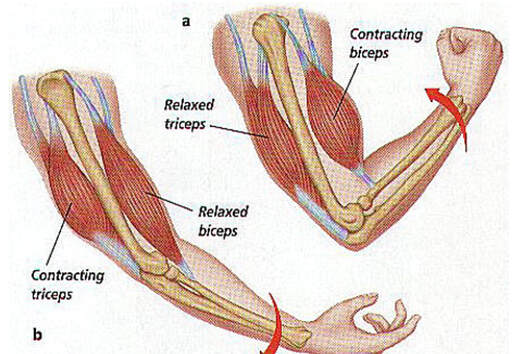
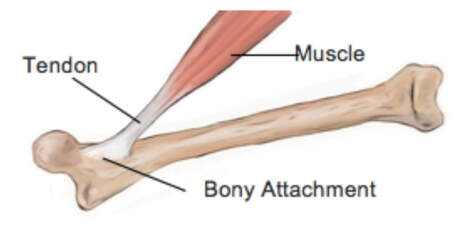
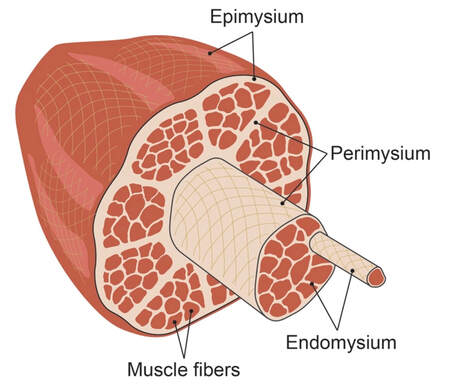
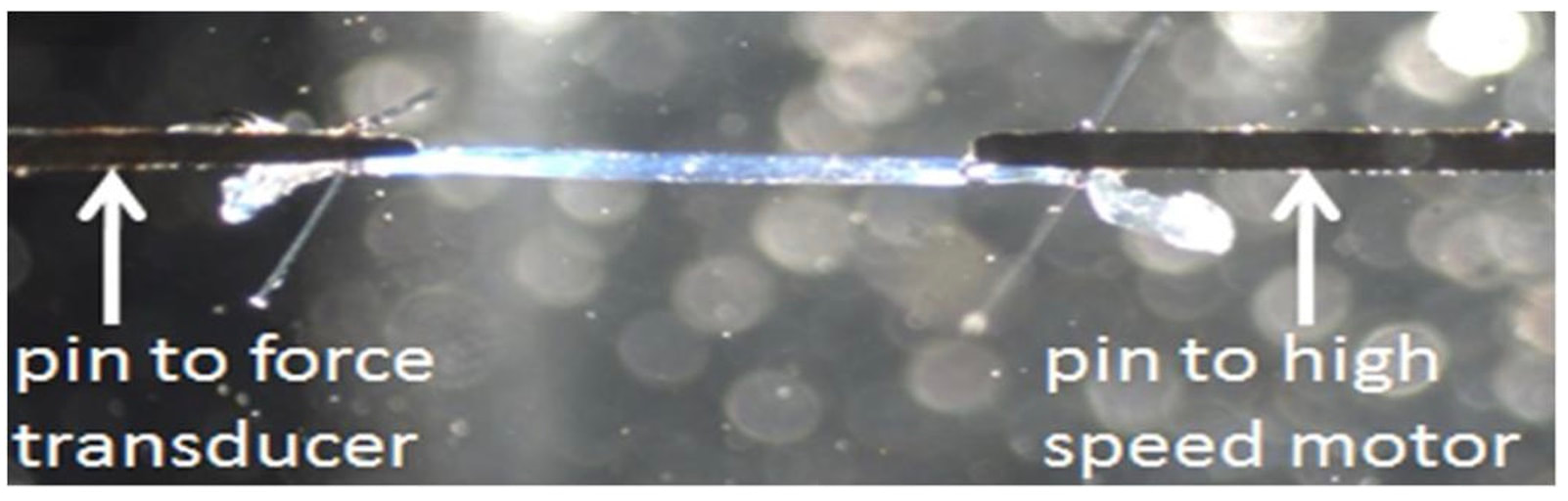
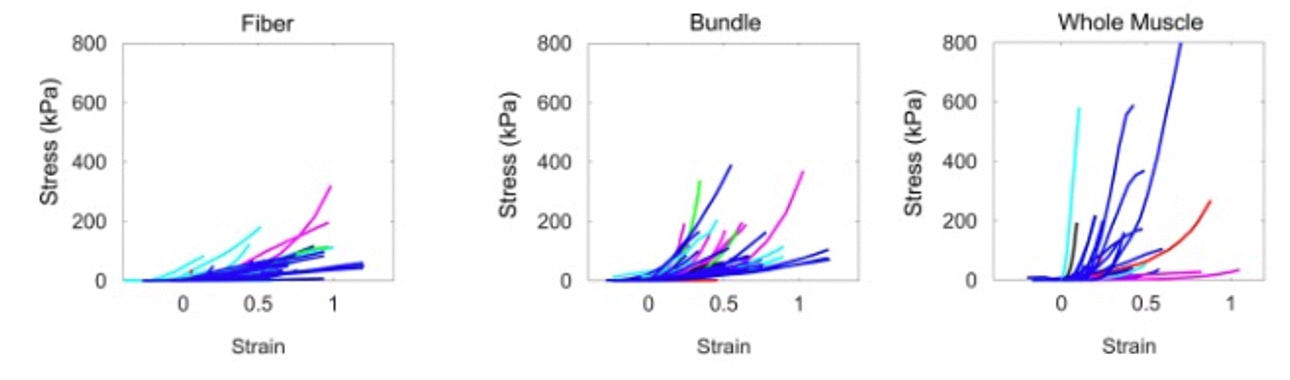
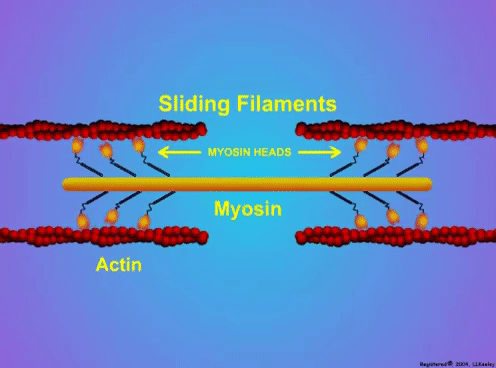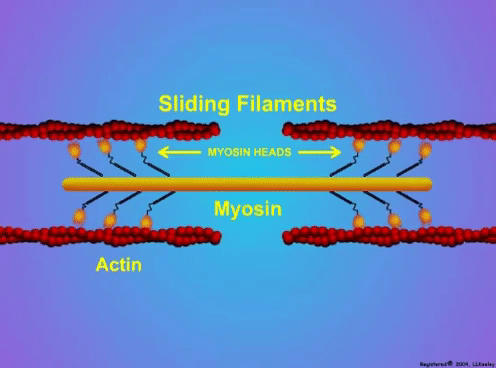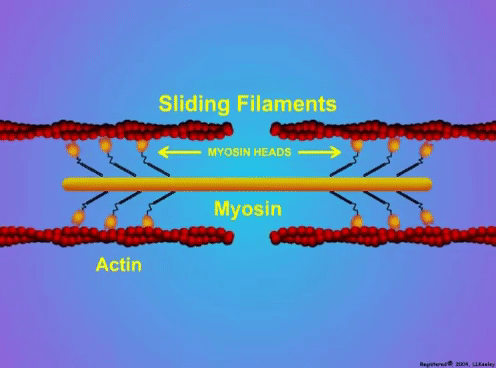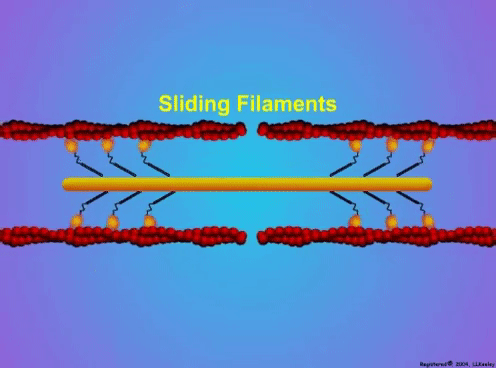
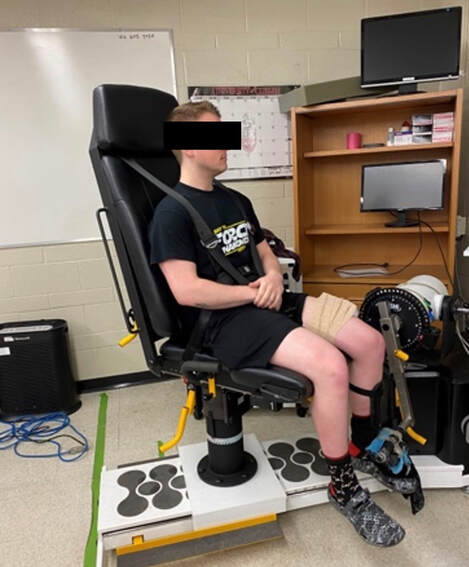
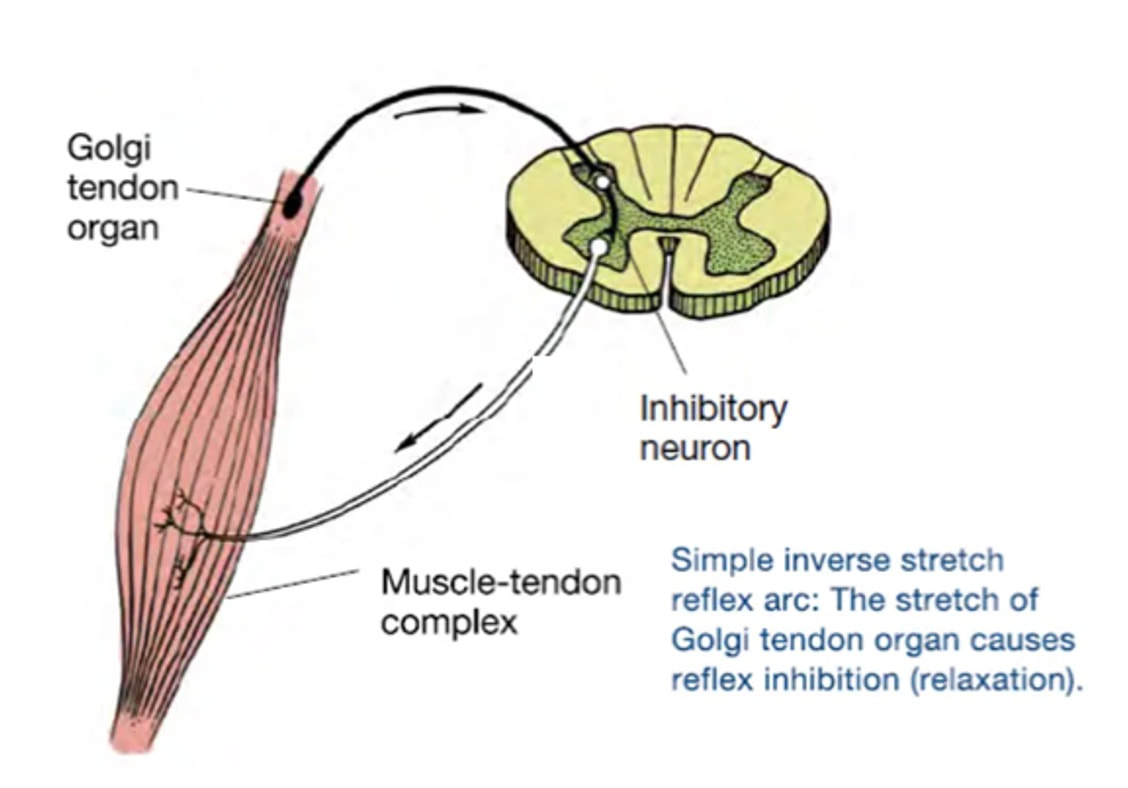
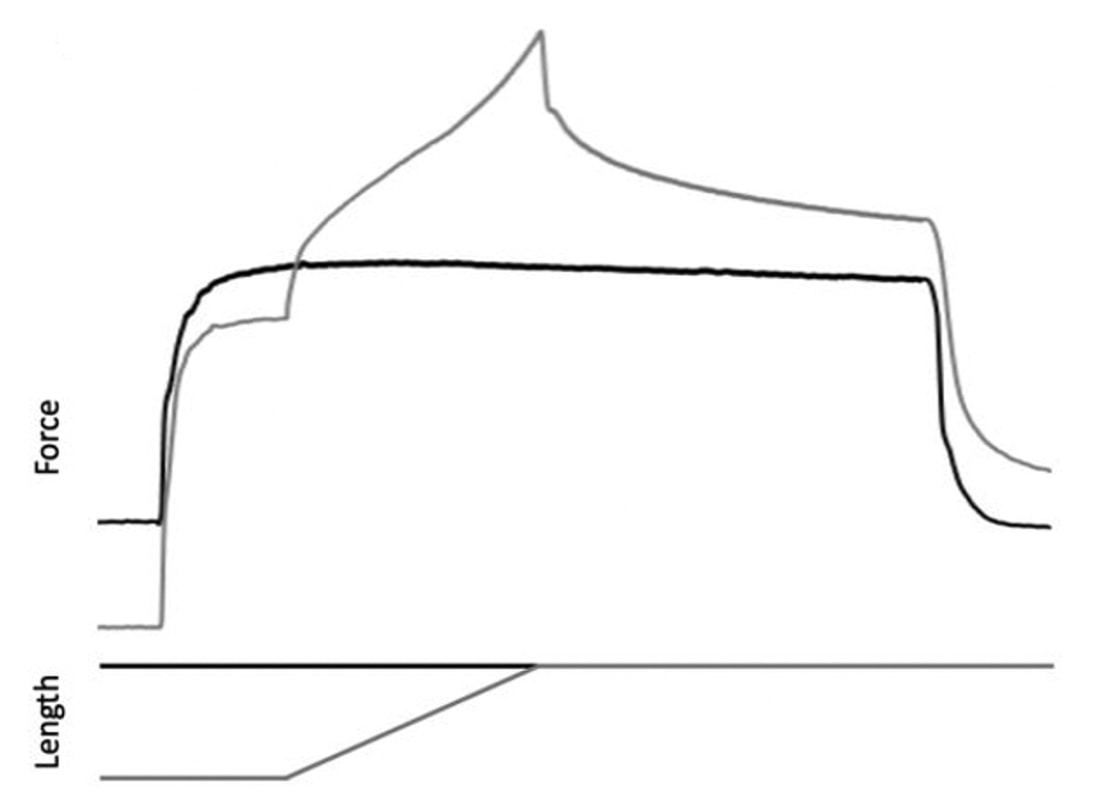
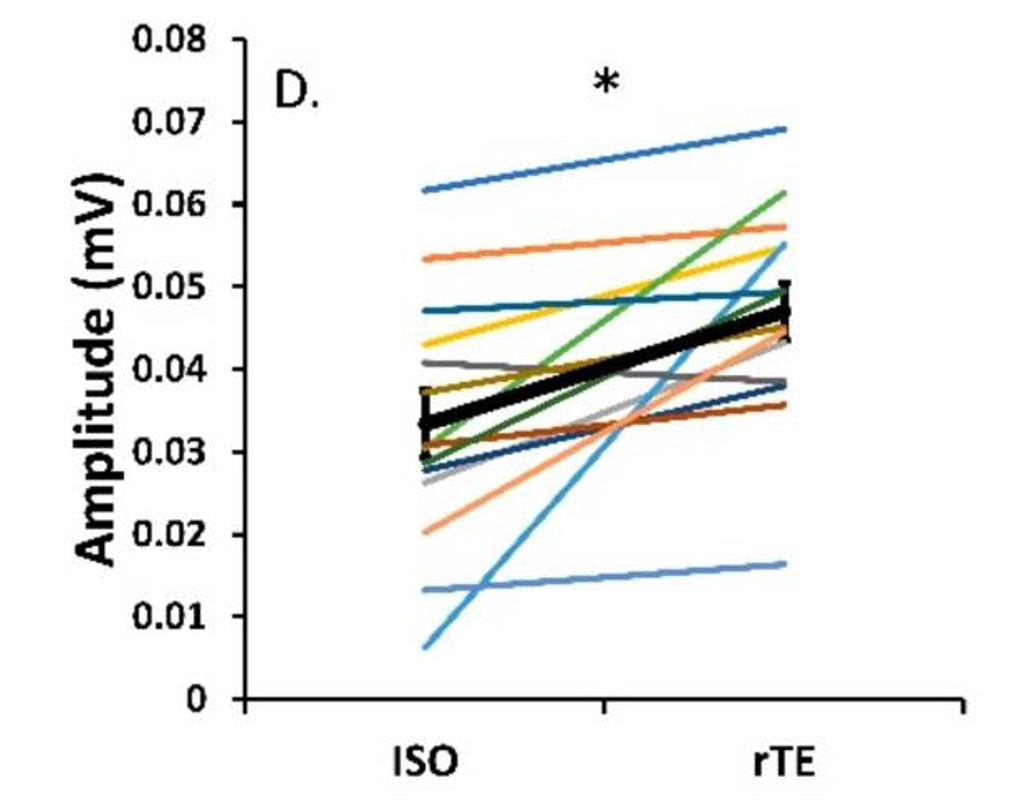
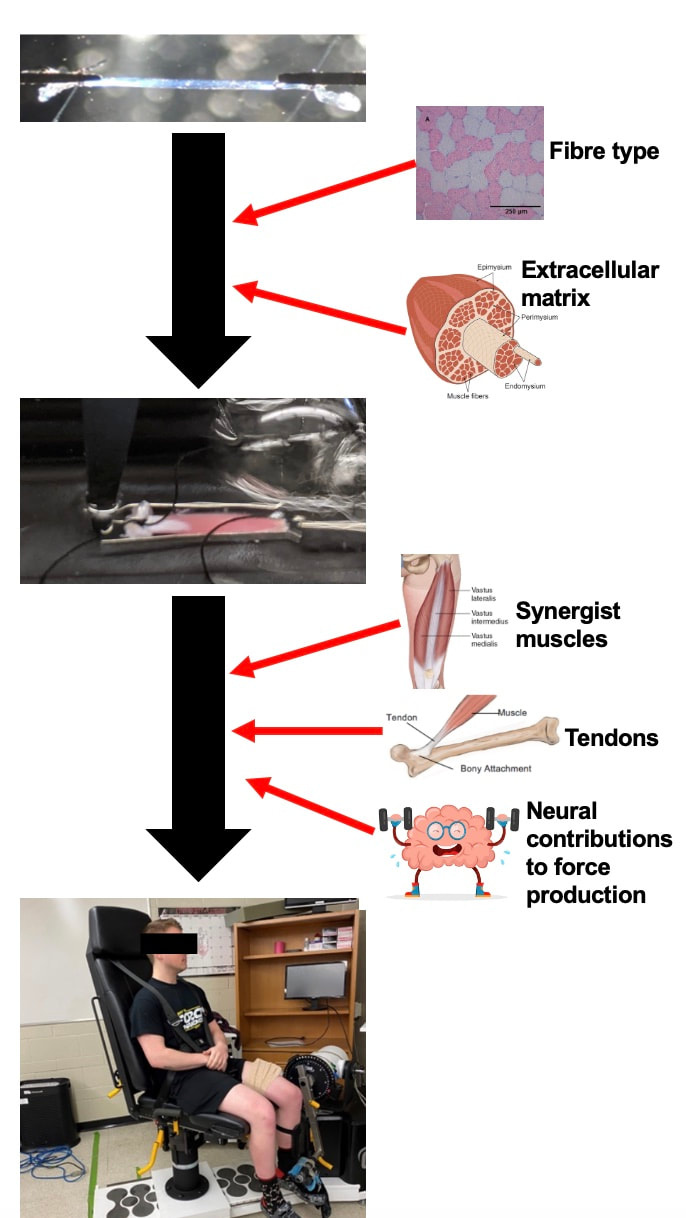
Can we scale muscle function from the single fibre to the joint level? (Insights from our lab)5/13/2023 By Avery Hinks In our lab, we measure muscle function in single fibres all the way up to whole groups of muscles producing movement together. We have a single fibre testing system in which we test fibres dissected from rodent muscles or human muscle biopsies. Our whole-muscle system allows us to test a whole dissected rodent muscle. On the largest scale, we can sit human participants in a chair called a “dynamometer” and instruct them to kick against a lever. At all scales, we perform similar assessments. These include maximum strength, maximum speed, how powerful a movement is—or just about any fundamental property of muscle function! We can do this because muscle structure is a predictable hierarchy. A whole muscle is made of many muscle fascicles, which are each made of many muscle fibres, which are each made of many myofibrils, which are each made of many sarcomeres aligned in series. Even with this hierarchy in mind, however, it might be hard to believe that what looks like a piece of a translucent noodle (that being a muscle fibre) functions the same as an entire group of muscles contracting to kick against a lever. And you’d be right to be skeptical! While the same biomechanical properties exist at all scales of muscle, there is rarely a one-to-one relationship from the single fibre to the whole muscle, and especially to the joint level. Why would this be the case, even when taking these measures within the same subject? In today’s knowledge translation, I will outline a few key considerations. Synergist muscles and fibre types When we produce any movement, it is rarely the result of just one muscle contracting. Kicking, for example, requires a coordinated effort among the four quadriceps muscles: vastus lateralis, rectus femoris, vastus intermedius, and vastus medialis. Even with this in mind, when attempting to equate muscle function from the single fibre to the joint level, we often focus solely on the vastus lateralis, ignoring the other quadriceps. But the vastus lateralis is the largest of the quadriceps muscles, and the easiest (and safest) to biopsy for single fibre experiments, so how much can we ignore those other muscles when comparing across scales? A study from our lab by Dalton and colleagues offers insight. In 8 young and 8 older men, they assessed rate of force development (a measure of how quickly one can develop maximum strength) in the whole quadriceps muscle group on a dynamometer and in vastus lateralis single fibres. In the figure below, the circled R^2 values on the larger graph correspond to the relationships indicated by red arrows on the smaller graphs. From these graphs, it is clear that single fibre rate of force development only related to joint-level rate of force development in slow-type fibres in old men. No other relationships existed between single fibre and joint-level rate of force development. Does this mean that single muscle fibre function and joint-level function are independent of one another? Definitely not. Based on the hierarchy of muscle structure, there would be no muscle without those fibres. What these graphs tell us is synergist muscles and other confounding factors discussed below provide enough of a disconnect between small-scale and large-scale muscle function that observing a clear relationship experimentally is difficult. The above graphs also raise a further consideration when going from the single fibre to the joint level: muscle fibre type. Most muscles consist of both “fast-twitch” and “slow-twitch” types of muscle fibres, the former being faster and stronger. In the above study, about 8 fibres were tested per person, but a whole vastus lateralis could contain hundreds of thousands of fibres. Perhaps 8 isn’t enough to represent the whole muscle, much less the whole joint, at the single fibre level. But what is enough? At the moment, our answer is as many as possible. We must also consider how time-consuming single fibre experiments can be, and the comfort of participants in giving multiple biopsies. Tendons and the extracellular matrix Muscles attach to bones, and as a muscle contracts, it draws two bones closer together, producing movement about a joint. But as you can see in the picture above, muscles do not attach directly to bone. Rather, muscle attaches to something that attaches to bone. That something is called a tendon. When a muscle contracts, it first pulls on the tendon, which then pulls on the bone it’s attached to, producing movement. If tendons were stiff like ropes, there would be no delay between when the muscle contracts and when the tendon pulls the bone. However, tendons are more like elastics, and undergo some initial stretch when muscles pull on them before that pull is transmitted to bone. Think of it like pulling a 10-pound weight with an elastic band—the weight will move eventually, but only after the elastic band has stretched a certain amount. Tendon stiffness varies from person to person, too. If a tendon is stiffer, there can be a closer relationship between shortening of the muscle and movement at the joint. If a tendon is more compliant (in other words, less stiff), there may be more initial muscle shortening before the joint moves. The same applies during muscle stretch: with a more compliant tendon, more of the stretch might go to the tendon, alleviating the amount of tension on the muscle itself. When we perform experiments on just one muscle, we can eliminate the tendon as a factor in muscle function. In our lab, this is only possible in rodents, after a muscle is dissected out, although recently labs in the United States also did this in humans intra-operatively. In the image below, we tied off the rat soleus muscle at the junctions where tendon and muscle meet, isolating the muscle from the tendons. What whole-muscle testing does not eliminate is connective tissue. Sheaths of connective tissue wrap around each scale of muscle discussed earlier. A sheath called the “epimysium” wraps around the whole muscle, a sheath called the “perimysium” wraps around each fascicle, and a sheath called the “endomysium” wraps around each fibre. Each of these sheaths also connect to one another, building a whole network called the “extracellular matrix.” Similar to the tendon, these sheaths of connective tissue transmit force when a muscle contracts—but between each muscle fibre and fascicle, and also to the tendon. If somebody has stiffer connective tissue, that will produce more tension in the muscle during stretch, even if the tendon has been removed from the equation. There are no sheaths of this connective tissue below the level of the single fibre. Therefore, single fibres allow us to assess muscle function without having to consider the impact of connective tissue. An excellent example of how connective tissue impacts muscle function from the fibre to the whole muscle can be found in a systematic review by Binder-Markey and colleagues. They compiled results across several studies that investigated the passive force-length relationship of muscle. The passive force-length relationship dictates, understandably, that as a muscle stretches, its tension increases. The graphs below show that this increase in tension becomes steeper when going from a single fibre to a fibre bundle to a whole muscle. Connective tissue is largely behind the greater steepness in this relationship when going from smaller to larger scales. Neural contribution to force production The primary basis for muscle force production exists in the “sliding filament theory.” When you activate a muscle, proteins called actin and myosin bind together to pull the Z-disc edges of a sarcomere (the smallest unit of a muscle) closer together. This process of actin and myosin binding dictates strength, speed, and power across all scales of muscle. There is another contributor to force production, though, that exists only in joint-level experiments: the brain. In our experiments on biopsied single muscle fibres or whole muscles dissected from a rat, we control every aspect of the muscle’s activation ourselves from a computer. When a participant sits in our dynamometer chair, however, and we tell them to kick against a lever, they control their muscle activation themselves. Sleep, stress, drugs, and numerous other factors can influence our brain’s performance. To activate a muscle, the brain sends a signal down the spinal cord, then along a nerve that connects directly to the muscle. These factors, in turn, can influence our muscle performance as well. Going a step further, the path from the brain to the muscle is not straightforward. Along the way, intersecting pathways can inhibit or further facilitate the signal from the brain. These inputs exist mainly at the brain and the spinal cord, and include inputs from pain sensors, sensors in the skin and joints, and sensors within the muscle being activated and other muscles. One example we’ve encountered in our lab is input from the “Golgi tendon organ” (GTO for short), a sensor that exists in tendons. The GTO senses stretch. When a tendon stretches past a certain point, the GTO sends a signal to the spinal cord to inhibit force production to protect the muscle. When we actively stretch a muscle, we can subsequently enhance its static force-producing ability. This phenomenon is called “residual force enhancement,” and is described further in a previous knowledge translation. We have observed residual force enhancement at every scale of muscle from the single fibre to the joint level. However, at the joint level, we sometimes encounter “non-responders,” people who do not exhibit residual force enhancement. By contrast, we ALWAYS see residual force enhancement in single fibres and dissected muscles. A study from our lab by Contento and colleagues observed a greater GTO reflex during residual force enhancement (“rTE” in the graphs below) compared to a reference contraction not preceded by active stretch (“ISO” in the graphs below). The GTO is far removed from the equation in our single fibre and whole-muscle experiments. Therefore, the non-responders we encounter at the joint level may be caused by the GTO’s inhibitory input or other inhibitory inputs at the spinal cord. ConclusionThe highly organized hierarchy of muscle structure makes equating muscle function from smaller to larger scales appealing. However, as we move to larger scales, we must increasingly consider confounding factors of tendons and the extracellular matrix, other muscles acting in synergy with the muscle of interest, and neural contributions to force production. So far, we have observed some instances of relationships between single fibre and joint-level function. Such observations may be easier to produce as we further refine techniques to account for these factors when attempting to match muscle function across scales. References
Binder-Markey BI, Sychowski D, Lieber RL. Systematic review of skeletal muscle passive mechanics experimental methodology. J Biomech. 2021 Dec 2;129:110839. doi: 10.1016/j.jbiomech.2021.110839. Epub 2021 Oct 26. PMID: 34736082; PMCID: PMC8671228. Binder-Markey BI, Persad LS, Shin AY, Litchy WJ, Kaufman KR, Lieber RL. Direct intraoperative measurement of isometric contractile properties in living human muscle. J Physiol. 2023 Mar 11. doi: 10.1113/JP284092. Epub ahead of print. PMID: 36905200. Chen J, Mashouri P, Fontyn S, Valvano M, Elliott-Mohamed S, Noonan AM, Brown SHM, Power GA. The influence of training-induced sarcomerogenesis on the history dependence of force. J Exp Biol. 2020 Aug 13;223(Pt 15):jeb218776. doi: 10.1242/jeb.218776. PMID: 32561632. Contento VS, Dalton BH, Power GA. The Inhibitory Tendon-Evoked Reflex Is Increased in the Torque-Enhanced State Following Active Lengthening Compared to a Purely Isometric Contraction. Brain Sci. 2019 Dec 23;10(1):13. doi: 10.3390/brainsci10010013. PMID: 31878094; PMCID: PMC7016668. Dalton BE, Mazara N, Debenham MIB, Zwambag DP, Noonan AM, Weersink E, Brown SHM, Power GA. The relationship between single muscle fibre and voluntary rate of force development in young and old males. Eur J Appl Physiol. 2023 Apr;123(4):821-832. doi: 10.1007/s00421-022-05111-1. Epub 2022 Dec 9. PMID: 36484861. Winkler T, Mersmann F, von Roth P, Dietrich R, Bierbaum S, Arampatzis A. Development of a Non-invasive Methodology for the Assessment of Muscle Fibre Composition. Front Physiol. 2019 Mar 11;10:174. doi: 10.3389/fphys.2019.00174. PMID: 30914961; PMCID: PMC6421337.
0 Comments
Your comment will be posted after it is approved.
Leave a Reply. |
AuthorAvery Hinks Archives
September 2023
Categories |







 RSS Feed
RSS Feed
