|
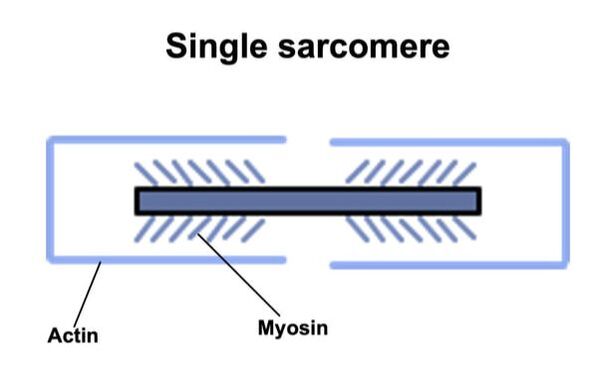
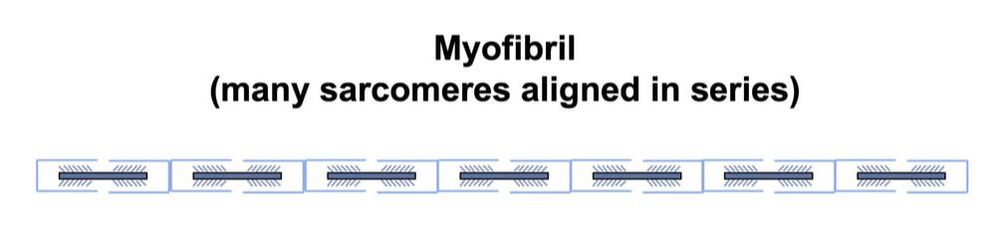
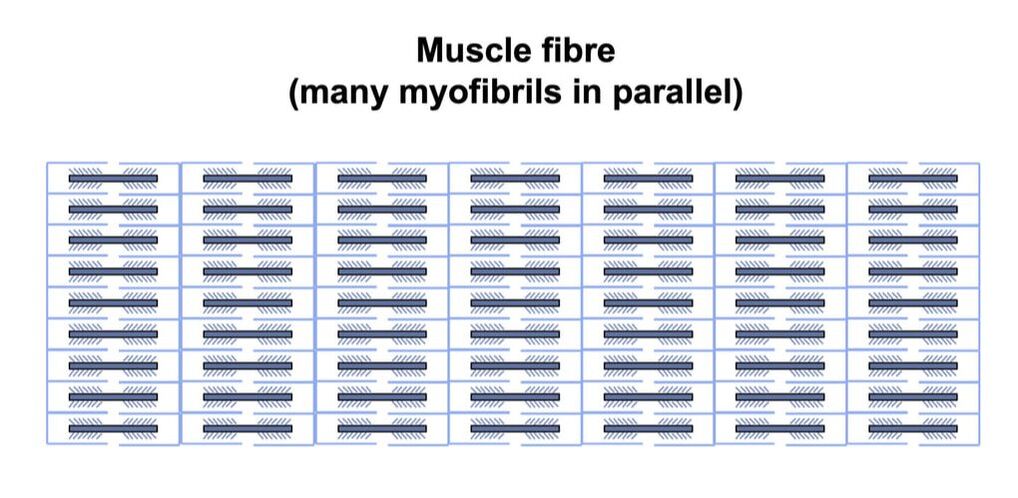
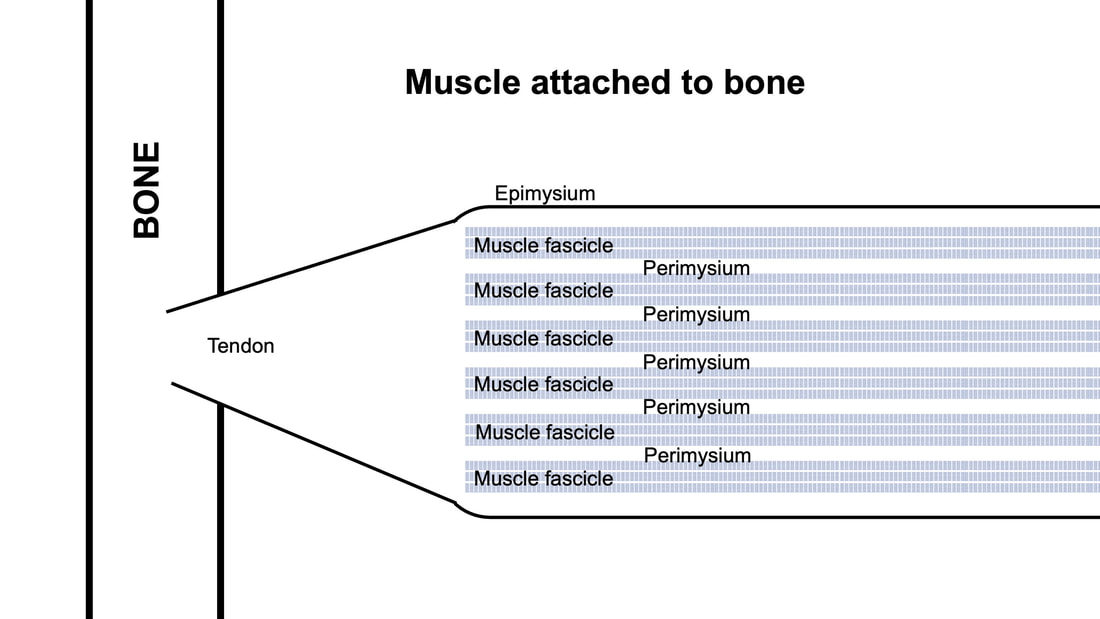
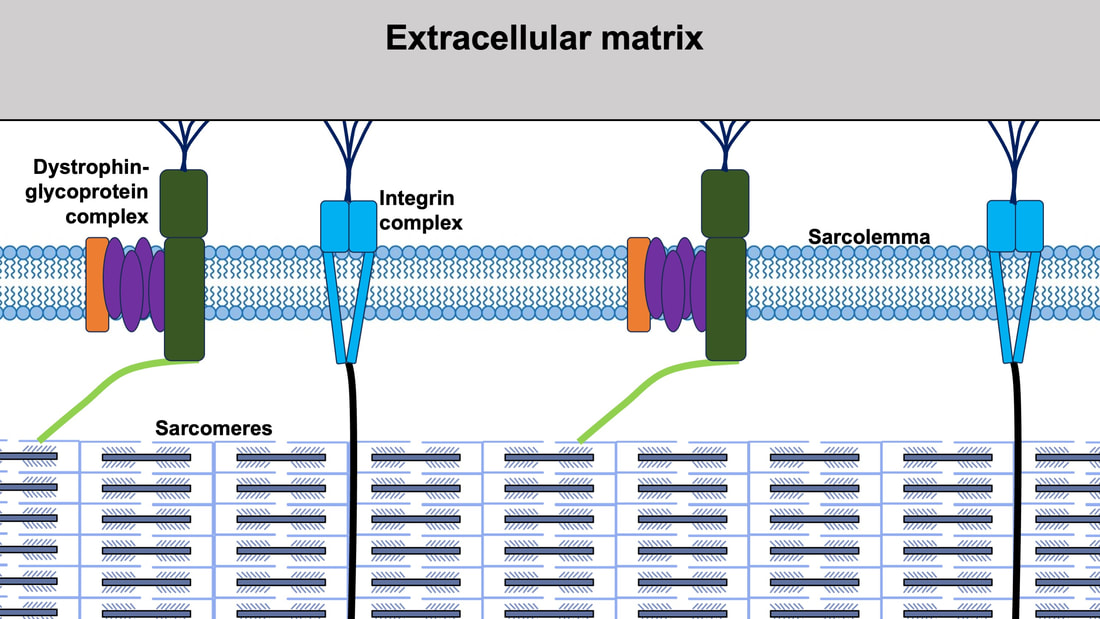
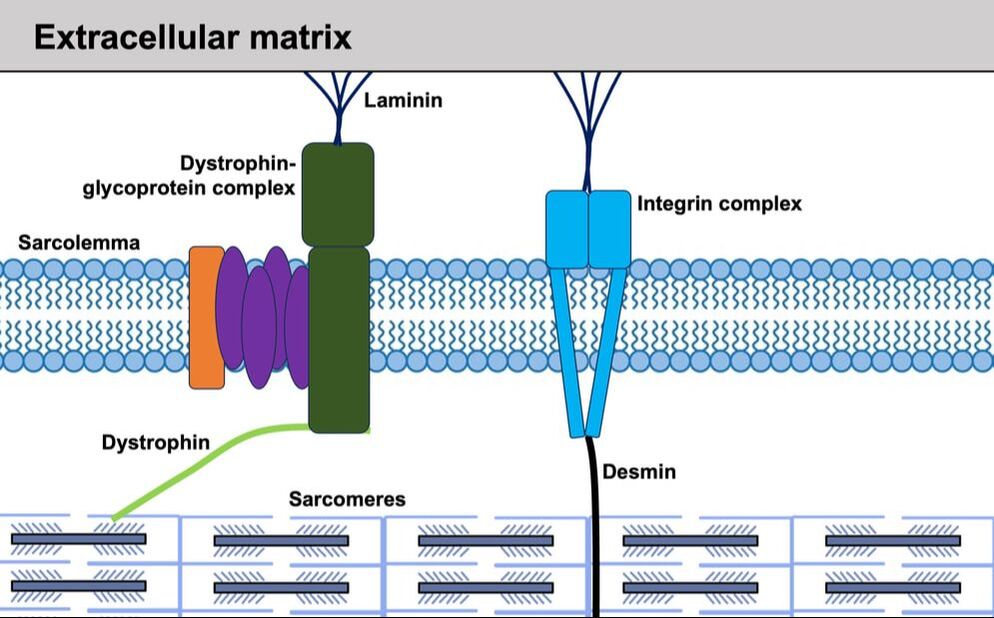
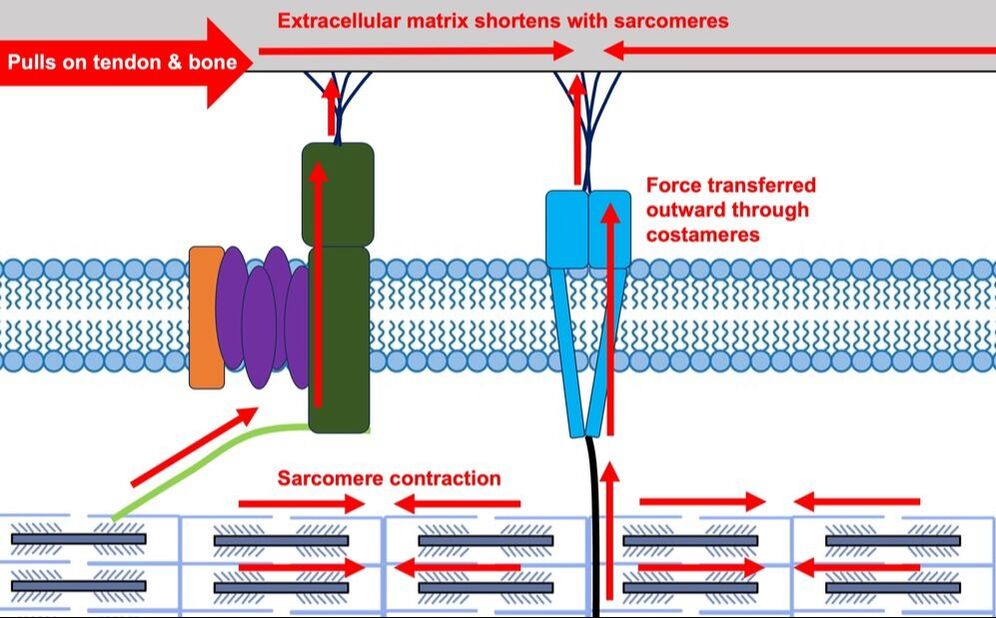
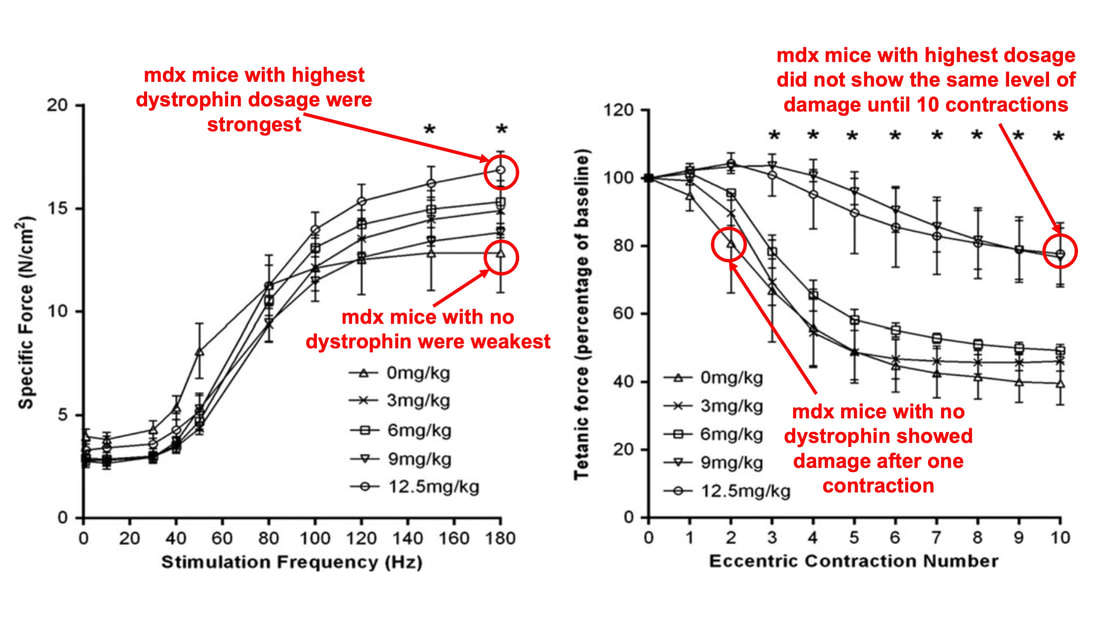
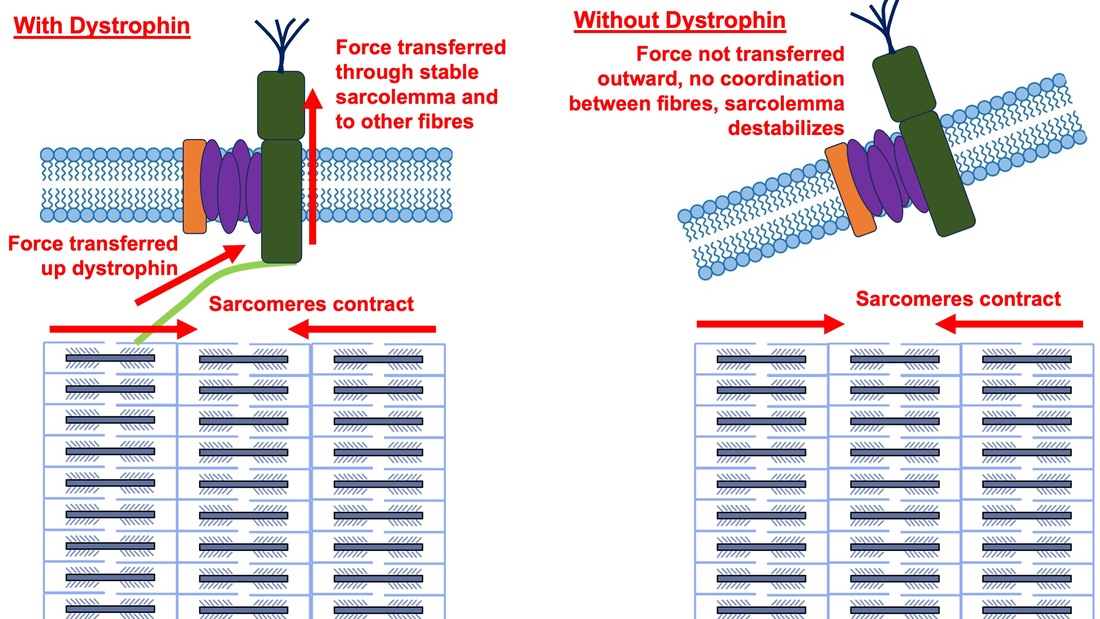
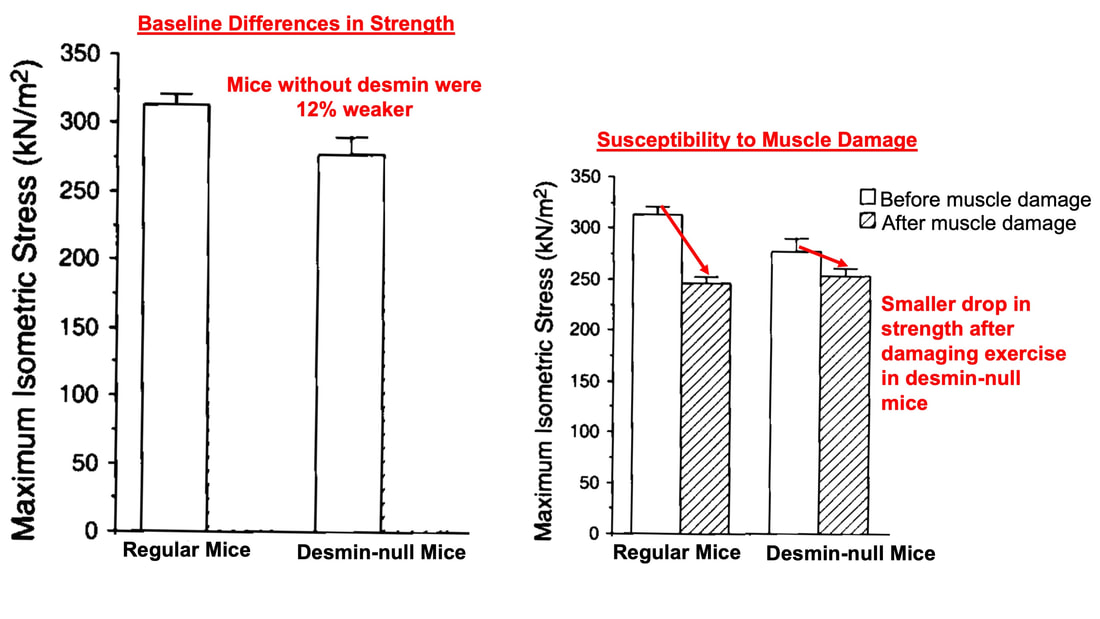
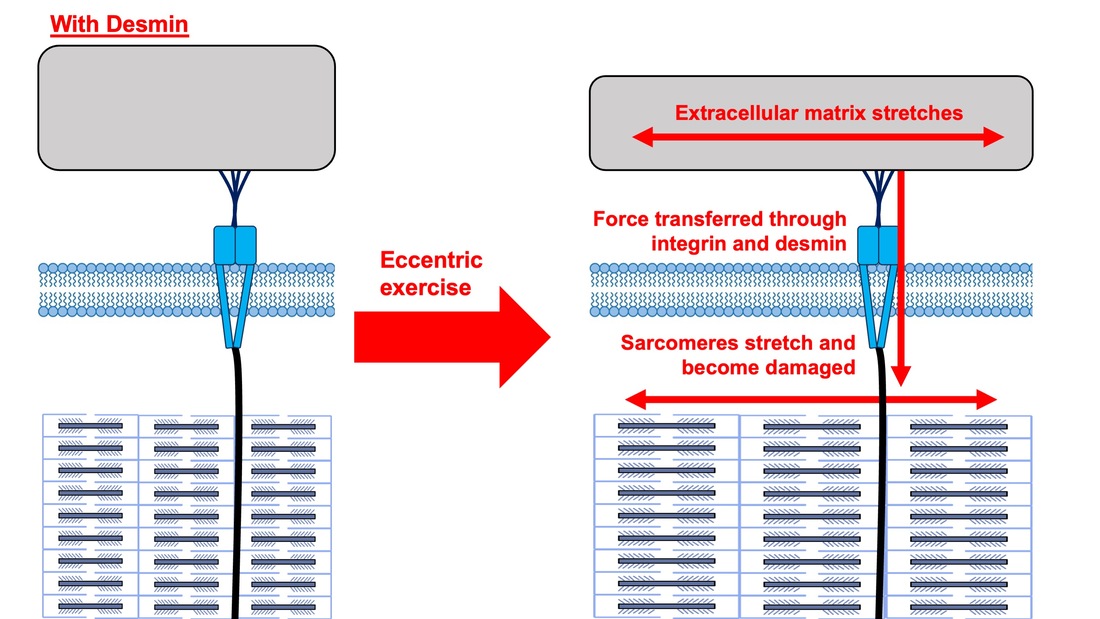
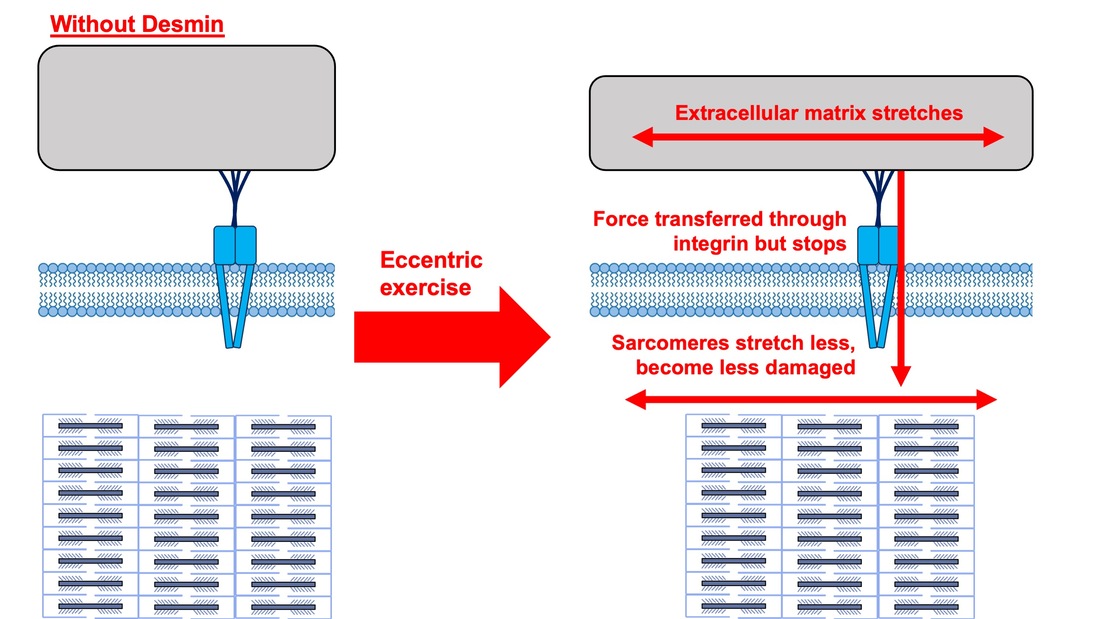
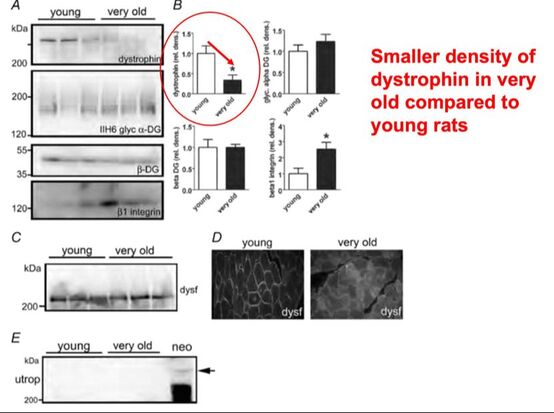
By Avery Hinks When we as physiologists describe how our muscles generate the force, we often refer to the smallest unit of muscle contraction, the sarcomere, and its proteins myosin and actin. A signal sent from the brain induces the release of calcium, which incites a series of events that cause actin and myosin proteins to bind, forming what we call “cross-bridges.” The formation of these cross-bridges in thousands of sarcomeres throughout a muscle produces the force that allows that muscle to lift, jump, and run. The thing is, there’s more going on between the formation of those cross-bridges and the movement of the muscle than we often acknowledge—steps in the process that are important but easy to overlook for purposes of simplicity. Here I ask the question: how are the forces created by those cross-bridges transmitted to the bone to produce movement? First, to get oriented…To put the above question in context, it’s important to understand how a muscle is organized from its smallest to largest scale. At the smallest level, we have the individual sarcomere, containing the force-generating actin and myosin filaments as described above. Each sarcomere is about 0.0025 millimetres long. Being so small, one sarcomere can’t do much alone, so thousands of sarcomeres aligned side by side comprise what’s called a myofibril. Within a muscle fibre, many of these myofibrils are packed parallel to one another. In humans, the diameter of a muscle fibre ranges from about 0.02-0.1 mm depending on how many myofibrils are packed together. So, at this scale we’re still small, but just visible to the human eye. Each muscle fibre is wrapped in a structure called the sarcolemma, which electrical signals travel down to tell the fibre to contract. The sarcolemma, in a way, separates each muscle fibre from one another. To further separate them, each muscle fibre is also surrounded by a sheath of collagen called the endomysium. Another, larger sheath of collagen called the perimysium surrounds many muscle fibres packed together. Each distinct bundle of muscle fibres wrapped in perimysium is called a muscle fascicle. Almost there. All of these muscle fascicles packed parallel to one another comprise the whole muscle, around which there is yet another layer of collagen called the epimysium. Collectively, these sheaths of collagen (endomysium, perimysium, epimysium) comprise the extracellular matrix. Lastly, the whole muscle needs to connect to bone, otherwise there’s no way for it to move our limbs. At each end of a muscle, the perimysium and epimysium merge into tendons, and those tendons attach directly to bones. You might’ve picked up on the problem here…if it’s the extracellular matrix that connects to bone, how the heck are the sarcomeres supposed to move bone? Wouldn’t that mean the muscle fibres just contract inside each of their own collagen sheaths without accomplishing anything? What connects sarcomeres to the extracellular matrix?Dispersed along the sarcolemma of each muscle fibre, there are large clusters of proteins called costameres. There are two types of costameres: the dystrophin-glycoprotein complex and the integrin complex. Other proteins extend off either end of these costameres to anchor them to both the sarcomere and the extracellular matrix. On the extracellular matrix side, this anchoring is done mostly by a protein called laminin. On the sarcomere side, there are several proteins we could focus on, but for the purposes of this article, I’ll focus on two: dystrophin and desmin. Laminin, dystrophin, desmin, and the proteins that makeup costameres are all classified as intermediate filaments. So, how is a sarcomere’s force transferred to the extracellular matrix? As the sarcomere contracts, it pulls on the intermediate filaments, then the intermediate filaments pull the extracellular matrix. As a result, force is transferred all the way from the sarcomere to the tendon then bone. Intermediate filaments also help coordinate movement between each muscle fibre. To understand what I mean by that, picture this like draft horses pulling weights at a fair. As you can see in the picture below, not only are the draft horses connected to the weights they’re pulling, they’re also connected to each other. Imagine the chaos that might ensue if one horse started moving faster than the other, or if they accidentally veered in opposite directions. That link (the intermediate filaments) between the horses (muscle fibres) keeps them moving in the same direction and speed as they drag those weights (bones). What if we didn’t have these intermediate filaments?Researchers have used mice lacking dystrophin (called “mdx mice”) to understand dystrophin’s importance for stabilizing muscle contractions. An impressive example came from Godfrey and colleagues at Oxford. They assessed muscle strength in mdx mice and the susceptibility to muscle damage during exercise. More importantly, they reintroduced dystrophin to mdx mice at varying levels of dosage to investigate whether that would improve their muscle function. As shown in the graphs below, mdx mice that did not receive any dystrophin treatment were the weakest (left graph) and the quickest to experience muscle damage during exercise (right graph). Unsurprisingly, introducing more and more dystrophin into their muscles progressively increased their strength and made them less susceptible to damage. When considering dystrophin’s role as described earlier, these results make sense. Dystrophin anchors the sarcomere’s actin filaments to the sarcolemma. Without dystrophin, there is less coordination between fibres, making some fibres more likely to be too compressed or stretched to produce maximal force. Additionally, the sarcolemma is left unstable, making it more likely to breakdown during intense exercise. Desmin also anchors the sarcomere to the sarcolemma, so it would be reasonable to expect a lack of desmin to induce effects similar to a lack of dystrophin…except it’s not quite the same. Sam and colleagues from the University of California performed an experiment similar to the dystrophin study described above, only using desmin-null mice (mice with a mutation causing them to not express desmin in their muscles). Like in mice lacking dystrophin, the researchers observed a 12% reduction in strength in desmin-null mice (left graph below). When exposing the muscles to intense exercise, however, the demisn-null mice actually experienced less damage than regular mice (right graph below). The loss of strength without desmin is the same as the argument for dystrophin: force isn’t as easily transferred between fibres, hindering the muscle’s ability to coordinate force production. Regarding damage, however, it seems that desmin’s different attachment points compared to dystrophin create an opposite effect. Desmin instead wraps around the Z-disks (i.e., the ends) of a sarcomere and connects to the integrin complex. Researchers have speculated that, without that connection between integrin and the Z-disks, the extracellular matrix bears more of the load during damaging exercise. As a result, the muscle fibres themselves are left less perturbed. This might seem like a benefit of desmin mutation, but an inability to damage muscle during exercise comes with disadvantages too. In order for a muscle to grow stronger following exercise, it needs to undergo some level of stress. If a lack of desmin prevents that from happening, the muscle is less likely to adapt to exercise. These studies are from genetically modified rodents. Why do we care?Do we, as humans, have to worry about the integrity of our intermediate filaments in the real world? While rare, there are in fact diseases that affect intermediate filaments. Duchenne’s muscular dystrophy affects 1 in 3500 males and causes muscle to lack dystrophin. Some people also experience desminopathy, which involves a mutation on desmin that makes it defective. Of greater concern, however, is that muscles lose dystrophin with age. Researchers at the University of Michigan showed that, like mdx mice, aged rats exhibit an impairment in the ability to transmit force between muscle fibres. That inability to transmit force was associated with a smaller density of dystrophin. So (surprise, surprise), loss of dystrophin is one of the many contributors to the loss of strength with age. Conclusion When discussing muscle contraction, focus is often placed on the sarcomere. While the sarcomere is of course important, its ability to translate force to bone and create movements would not be possible without the intermediate filaments that anchor it to the sarcolemma and extracellular matrix. Without intermediate filaments, like draft horses with no reins binding them together, chaos ensues.
References Godfrey C, Muses S, McClorey G, Wells KE, Coursindel T, Terry RL, Betts C, Hammond S, O'Donovan L, Hildyard J, El Andaloussi S, Gait MJ, Wood MJ, Wells DJ. How much dystrophin is enough: the physiological consequences of different levels of dystrophin in the mdx mouse. Hum Mol Genet. 2015 Aug 1;24(15):4225-37. doi: 10.1093/hmg/ddv155. Epub 2015 May 1. PMID: 25935000; PMCID: PMC4492390. Ramaswamy KS, Palmer ML, van der Meulen JH, Renoux A, Kostrominova TY, Michele DE, Faulkner JA. Lateral transmission of force is impaired in skeletal muscles of dystrophic mice and very old rats. J Physiol. 2011 Mar 1;589(Pt 5):1195-208. doi: 10.1113/jphysiol.2010.201921. Epub 2011 Jan 10. PMID: 21224224; PMCID: PMC3060596. Sam M, Shah S, Fridén J, Milner DJ, Capetanaki Y, Lieber RL. Desmin knockout muscles generate lower stress and are less vulnerable to injury compared with wild-type muscles. Am J Physiol Cell Physiol. 2000 Oct;279(4):C1116-22. doi: 10.1152/ajpcell.2000.279.4.C1116. PMID: 11003592.
0 Comments
Your comment will be posted after it is approved.
Leave a Reply. |
AuthorAvery Hinks Archives
September 2023
Categories |




 RSS Feed
RSS Feed
