|
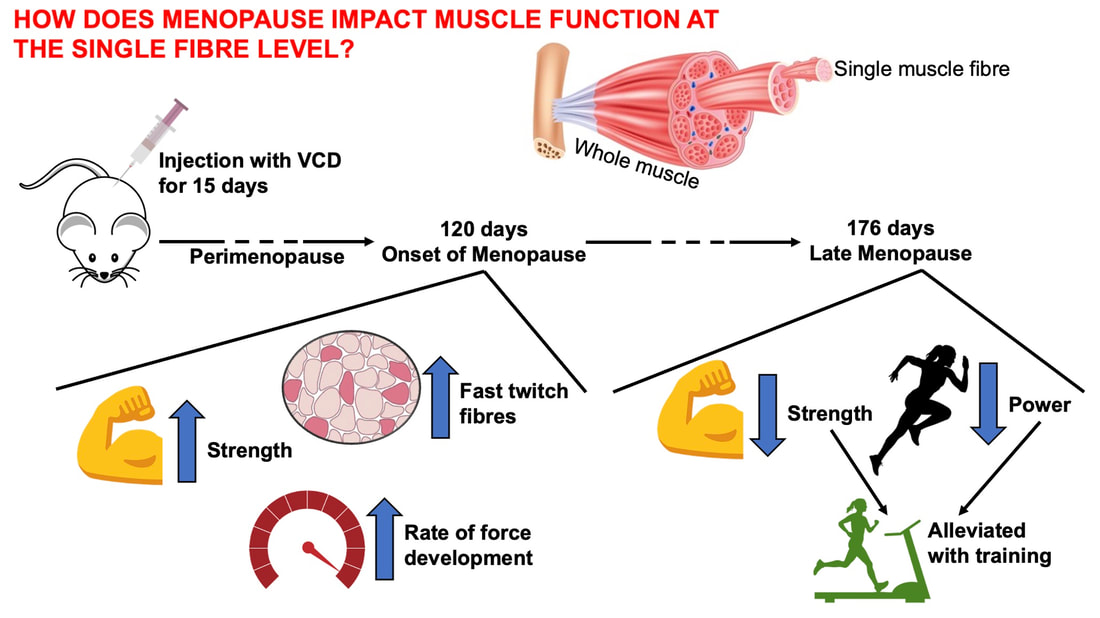
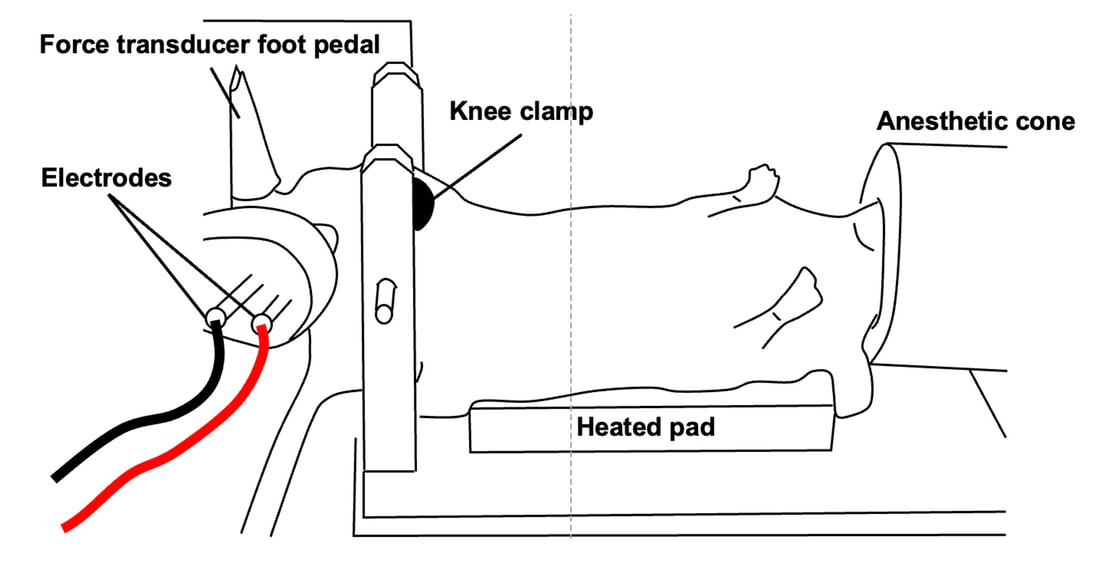
By Avery Hinks Around age 40 or 50, women experience a reduction in ovarian hormones leading to the cessation of menstruation—an event termed “menopause.” This event is not only associated with changes in the ovaries, however. The hormonal changes correspond to changes in various systems throughout the body including the cardiovascular system, central nervous system, musculoskeletal system, and more. In humans, muscle strength progressively declines into old age. This decline becomes more drastic in women than men at around the age of 50. Researchers have understandably pointed to menopause as a contributor to this sex difference in the age-related decline of strength. However, our understanding of what goes on in muscle during menopause is limited. Studies on mice have looked at the above question more closely by using an ovariectomized model. In these mice, either one or both of the ovaries are surgically removed, leaving them with no ovarian hormones. These mice allow researchers to then assess how muscle function changes in the absence of ovarian hormones compared to mice in which the ovaries were left intact. The ovariectomized model has a few shortcomings, however. Most importantly, ovariectomized mice have ovarian hormones one day, then suddenly don’t the next day. That is not how menopause works. A transitional period called “perimenopause” precedes actual menopause, marked by a gradual reduction in ovarian hormones. In humans, this gradual reduction occurs on average over 3-4 years, but can sometimes last up to a decade. The ovaries also retain the ability to produce testosterone during perimenopause, which would be missed by the ovariectomy model’s complete removal of the ovaries. A more recent model employing the occupational chemical 4-vinylcyclohexene diepoxide (thankfully shortened to just “VCD”) aims to better mimic the gradual reduction in ovarian hormones that occurs during natural menopause. This model allows for investigation of how muscle function changes both leading up to and following the onset of menopause. Our lab has so far published two studies using the VCD model. Here’s what we’ve found so far… The influence of a mouse model of perimenopause on muscle fibre functionOur lab’s recent paper by Mashouri and colleagues induced gradual ovarian failure in mice by VCD injection. We then assessed muscle fibre function 120 days after the injections, which corresponds to the onset of menopause. These assessments involve dissecting out a single fibre from a mouse muscle then tying it to a system that can control its length and activation (pictured below). Altogether, we could investigate how perimenopause impacts performance at one of the smallest levels of muscle contraction. In this study, we investigated four main measures of muscle fibre function:
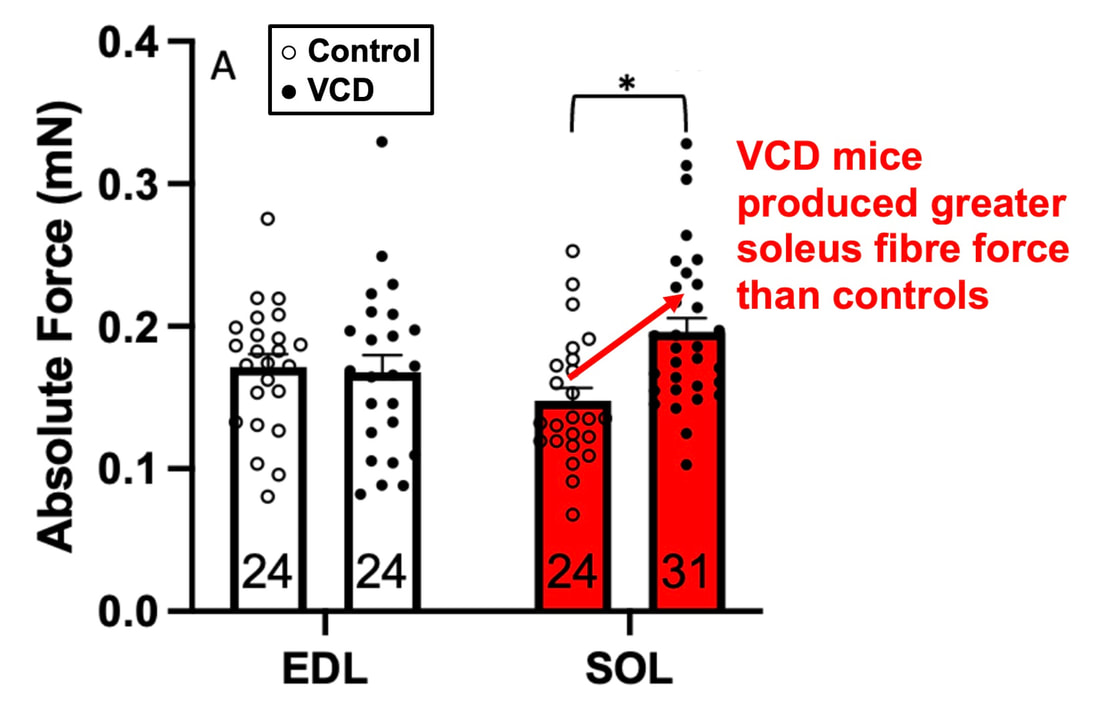
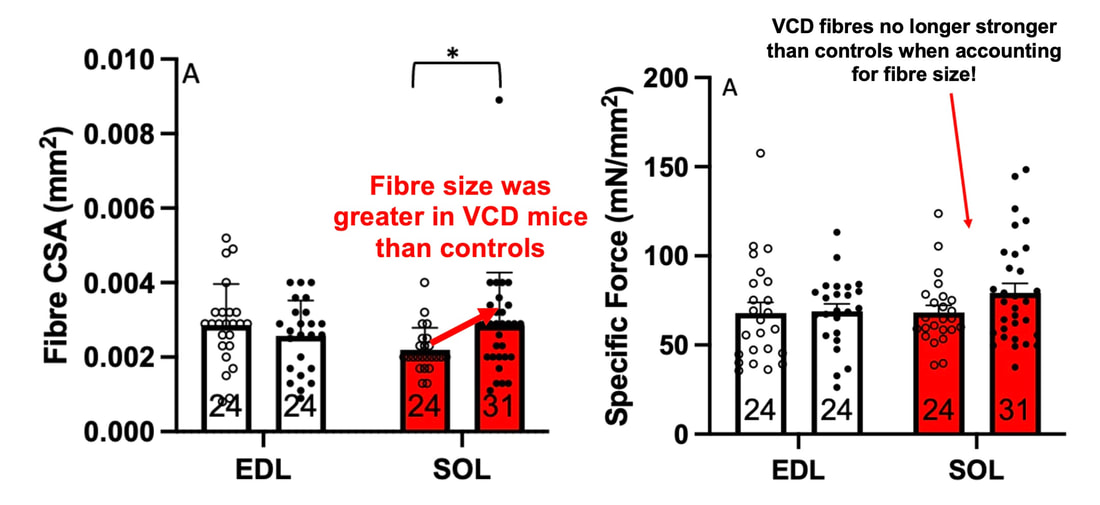
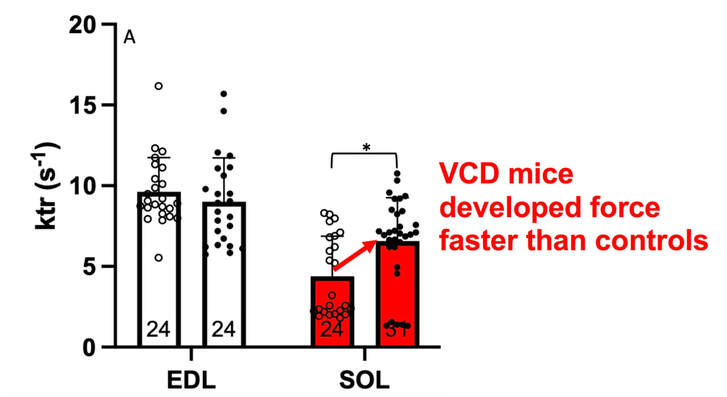
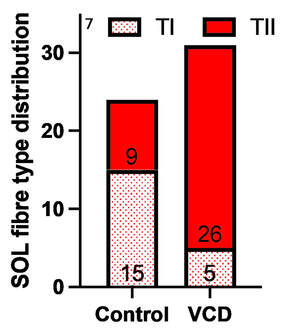
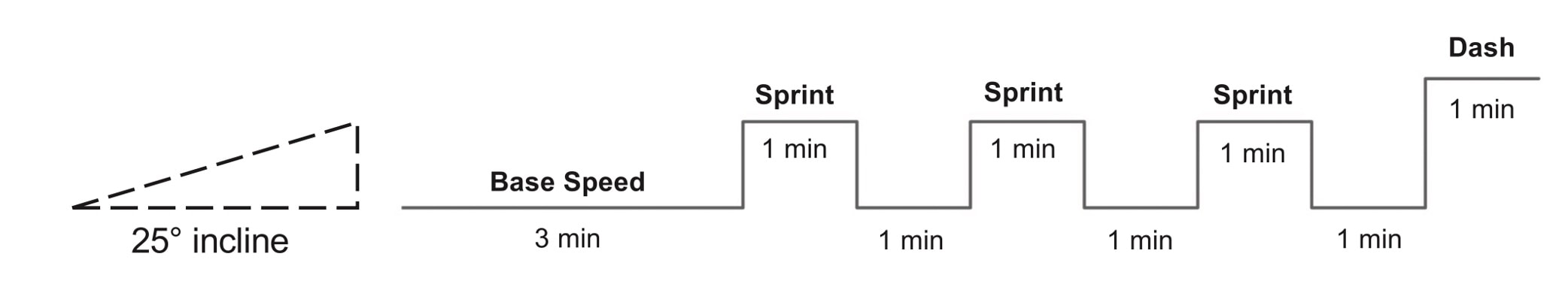
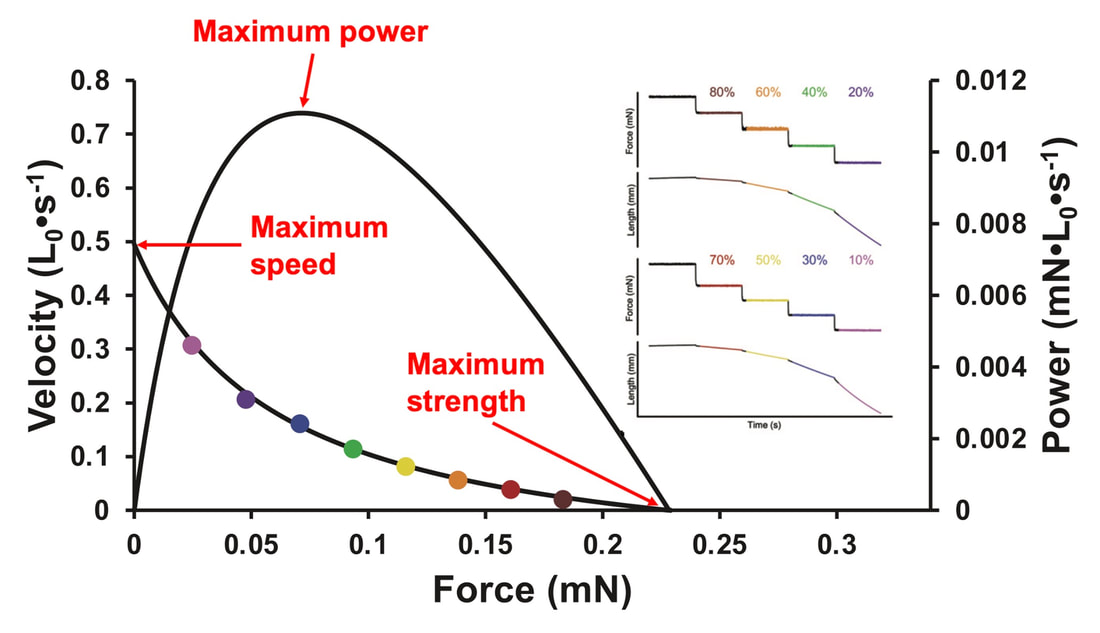
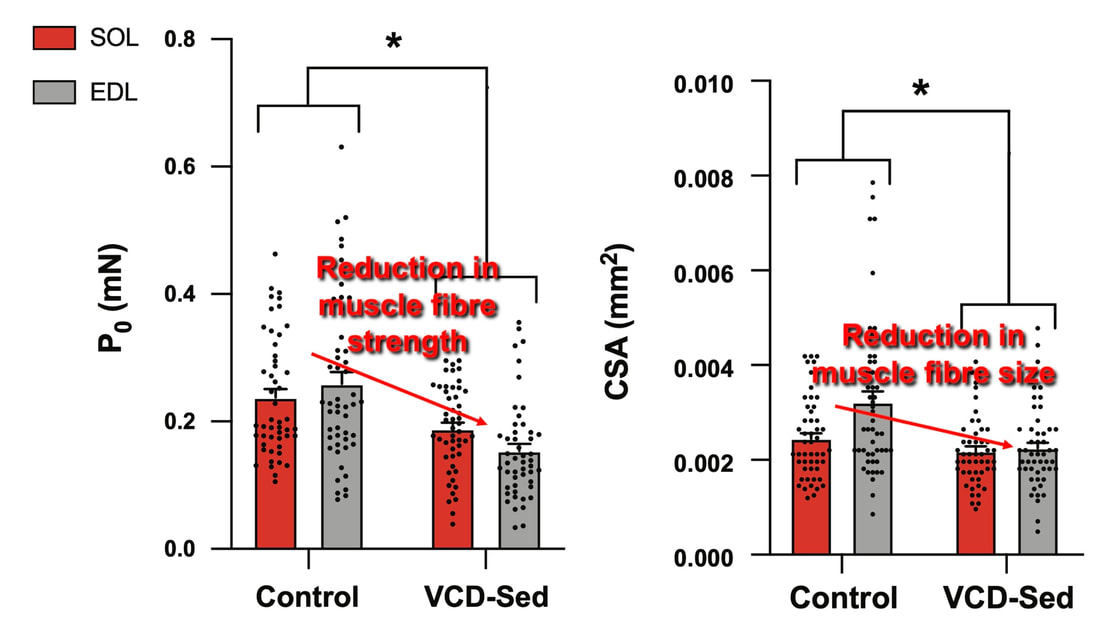
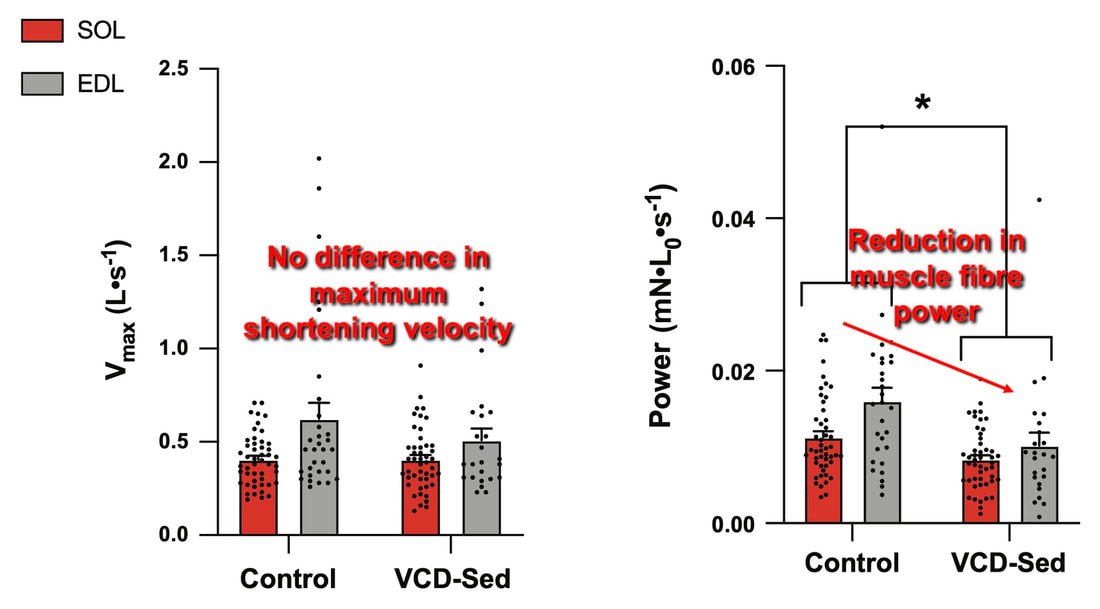
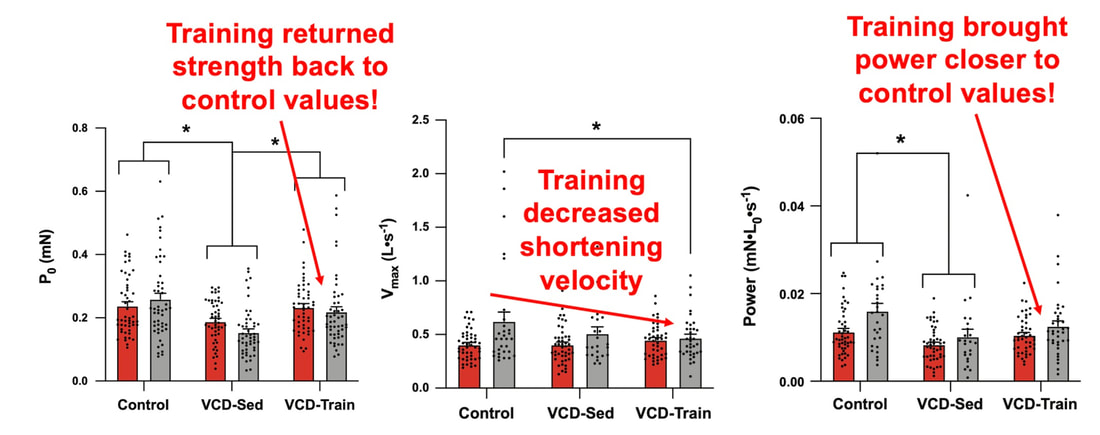
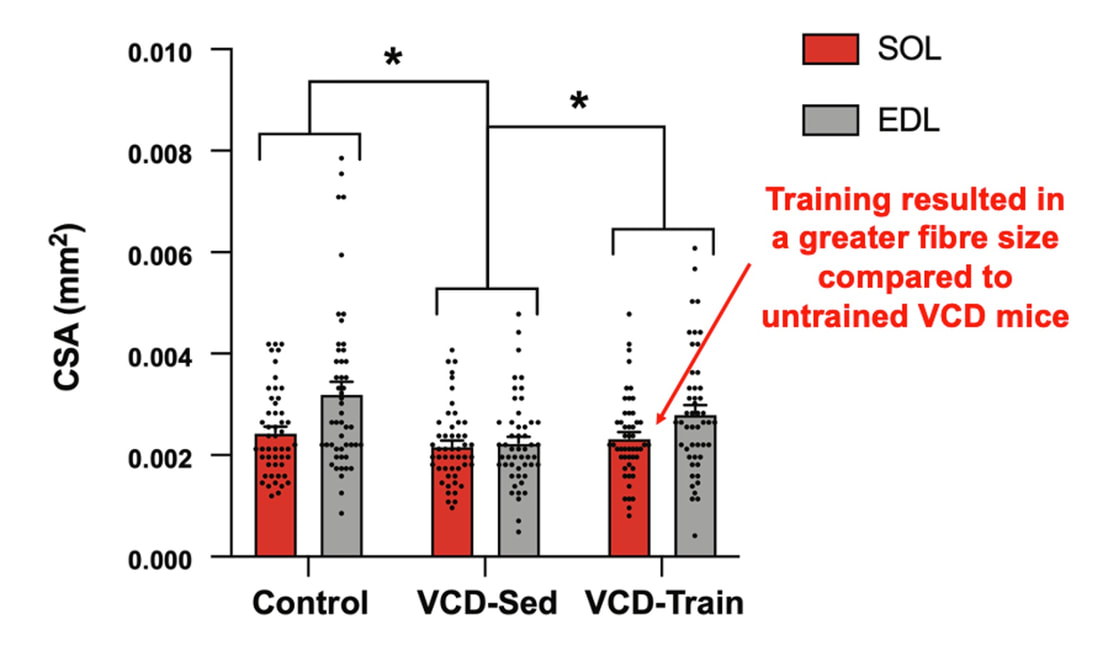
The graph below shows differences in absolute force between control and VCD mice. The muscles assessed were the extensor digitorum longus (EDL) and soleus (SOL), which are located on the shin and are responsible for pointing the toes. Surprisingly, at the onset of menopause, VCD mice had greater soleus single fibre force than controls! You might be thinking this goes against what we would expect with menopause. It certainly does, however, looking deeper provided clarity. Like with absolute force, we saw that soleus fibre cross-sectional area (“CSA” in the graph below) was also greater in VCD than control mice. Furthermore, normalizing absolute force to cross-sectional area (“specific force” in the graph below) eliminated the difference in force between control and VCD mice. These findings tell us that the greater force in VCD soleus fibres was largely due to a greater fibre cross-sectional area. We also found that soleus fibres of VCD mice could develop force faster than controls. These, again, must seem like the exact opposite of what we would expect with menopause. We must consider from these data that the single fibres are being tested alone, and do not necessarily represent how the muscle would function as a whole. In particular, muscle fibres come in two types: type I and type II. Type I muscle fibres are smaller, slower, and weaker. Type II muscle fibres, conversely, are larger, faster, and stronger. Therefore, perhaps these findings are indicative of VCD mice having more type II muscle fibres… And that is precisely what we found. As shown in the graph below, the soleus muscle of VCD mice had a larger proportion of type II fibres than controls! So, what does all this tell us? During perimenopause, gradual ovarian failure may cause a shift toward a greater proportion of type II muscle fibres in the soleus. This shift manifests as greater strength, size, and rate of force development within individual muscle fibres. We speculated that this shift may represent a compensatory adaptation in preparation for the declines in function that are about to occur with menopause. As well, type II fibres are more fatigable than type I fibres. Therefore, this fibre type shift could have a negative impact on muscle function in the form of greater fatigability. While this study provided important insight into how muscle function changes during perimenopause, two questions remained unanswered. First, this study only investigated the onset of menopause, leaving findings on what happens during menopause unanswered. Second, this study only investigated static measures of muscle fibre performance. The effect of ovarian failure on muscle function during movement was therefore unknown. Our next study addressed both of these questions. The influence of VCD-induced ovarian failure on muscle fibre dynamic contractile functionA study from our lab by Hubbard and colleagues used the same model as the study above. This time, however, mice were assessed 176 days after VCD injections. The onset of ovarian failure occurs at 120 days in these mice, therefore, 176 days (8 weeks later) is viewed as “late menopause.” This study also introduced high intensity interval training as a factor. High intensity interval training (“HITT” for short) involves brief but intense bursts of exercise separated by brief rests. For these mice, the type of HITT exercise was uphill running, and proceeded in the order shown below. A group of mice began training 3 days/week 120 days after VCD injections. In other words, we wanted to see if training starting at the onset of menopause could mitigate any impairments in muscle function observed in late menopause. This time, in addition to assessing the muscle fibres’ maximum strength, we also assessed maximum speed (known as “maximum shortening velocity”). However, a muscle’s force and velocity are inversely related—meaning that as a muscle shortens faster, the possible force it can produce decreases. We can combine force and speed together by assessing a muscle fibre’s maximum power, which is equal to force multiplied by velocity. In the graph above, you’ll see that maximum power occurs when both force and velocity are not maximal. This trade-off between force and velocity makes power the gold standard for assessing a muscle’s function during movement. Power is the kind of measure you'd use to assess someone's performance in jumping as high as they can. Hubbard and colleagues saw that late into menopause, muscle fibres were 30% deficient in strength (“Po” in the graph below). This reduction in strength coincided with similar reductions in muscle fibre size (“CSA” in the graph below). Notably, these results are different from what Mashouri and colleagues observed at the onset of menopause. Therefore, the progression of menopause, but not necessarily the phase of perimenopause leading up to it, is characterized by a reduced capacity for muscle fibres to generate strength. Interestingly, we saw no difference in the maximum shortening velocity of muscle fibres (Vmax in the graph below) between control and sedentary VCD mice. However, we still observed a 30% reduction in the fibres’ maximum power. Combined with the findings above, we can gather that a reduction in dynamic performance with menopause stems from a reduction in muscle fibre strength but not speed. But the big question is: did high intensity interval training mitigate the loss of strength and power? The graphs below show a comparison between control mice, sedentary VCD mice (VCD-Sed) and trained VCD mice (VCD-Train) for strength (Po), maximum shortening velocity, and maximum power. Training maintained the strength of VCD muscle fibres up to the values of control mice (shown on the left below). The effect of training on maximum power was not quite as strong, however, training still increased power by 20% compared to untrained VCD mice (shown on the right below). As mentioned above, power encompasses both force and velocity. Therefore, the smaller effect of training on power seems to be due to training having no beneficial effect on maximum shortening velocity (shown in the middle below). Additionally, muscle fibres of trained VCD mice had a greater cross-sectional area than untrained VCD mice. This maintenance of fibre size with training likely accounted for some of the maintenance of strength. Overall, this study told us that:
Conclusion and Next StepsAssessments of menopause in animals have historically employed the ovariectomy model, which results in a sudden elimination of ovarian hormones. The natural trajectory of menopause is more gradual, including a transition phase of perimenopause. By instead inducing gradual ovarian failure in mice, the VCD model has allowed us to gain a better understanding of how muscle fibre function changes leading up to and following menopause. We now know that during perimenopause, there is a shift toward a greater proportion of fast twitch muscle fibres, which manifests as fibres being larger, producing greater force, and producing force more rapidly. This fibre type shift may be compensatory, because later into menopause, we see reductions in fibre size, force, and power. High intensity interval training at the onset of menopause, however, can alleviate those impairments in muscle fibre function. These findings were at the level of the muscle fibre. Therefore, we do not know how the effects of menopause on muscle fibre function would translate to the performance of several muscles surrounding a joint. Those assessments, of course, could be more applicable to everyday life. Our lab is currently looking into that exact topic, repeating the above experiments on mice for joint-level assessments of muscle performance (pictured below). References
Mashorui P, Saboune J, Pyle WG, Power GA. Effects of chemically induced ovarian failure on single muscle fiber contractility in a mouse model of menopause. Maturitas. 2023 November 11. https://doi.org/10.1016/j.maturitas.2023.107885 Hubbard EF, Mashouri P, Pyle WG, Power GA. The effect of gradual ovarian failure on dynamic muscle function and the role of high-intensity interval training on mitigating impairments. Am J Physiol Cell Physiol. 2023 Oct 1;325(4):C1031-C1045. doi: 10.1152/ajpcell.00318.2023. Epub 2023 Sep 4. PMID: 37661923.
0 Comments
Your comment will be posted after it is approved.
Leave a Reply. |
AuthorAvery Hinks Archives
September 2023
Categories |



 RSS Feed
RSS Feed
